What research qualifies as exempt?
“Exempt” research are human subjects studies that present no greater than minimal risk to subjects and fit into one or more exempt categories (as described below).
What is exempt category?
To be classified as exempt, the research: Must involve only procedures or be a type of research study listed in one or more of the exempt categories (see exempt categories sections below); The study cannot involve prisoners as research subjects. Cannot be greater than minimal risk.
Is informed consent required for exempt research?
The HHS regulations at 45 CFR part 46 for the protection of human subjects in research require that an investigator obtain the legally effective informed consent of the subject or the subject’s legally authorized representative, unless (1) the research is exempt under 45 CFR 46.101(b); (2) the IRB finds and documents …
Who has the responsibility to determine whether a proposed project is exempt from review by the IRB?
The IRB reviewer determines if the proposed research is exempt from federal policies governing human subject protections. This determination is made in accordance with: The OHRP Decision Chart # 2- “Is the Research Involving Human Subjects Eligible for Exemption under 45 CFR 46.104(d)?”
What does it mean to be IRB exempt?
Research can be approved as “exempt” if it is no more than “minimal risk” and fits one of the exempt review categories as defined by federal regulation 45 CFR 46. Studies that may qualify for “Exempt” must be submitted to the IRB for review. Exempt reviews are conducted by a member of IRB staff.
What is exempt human research?
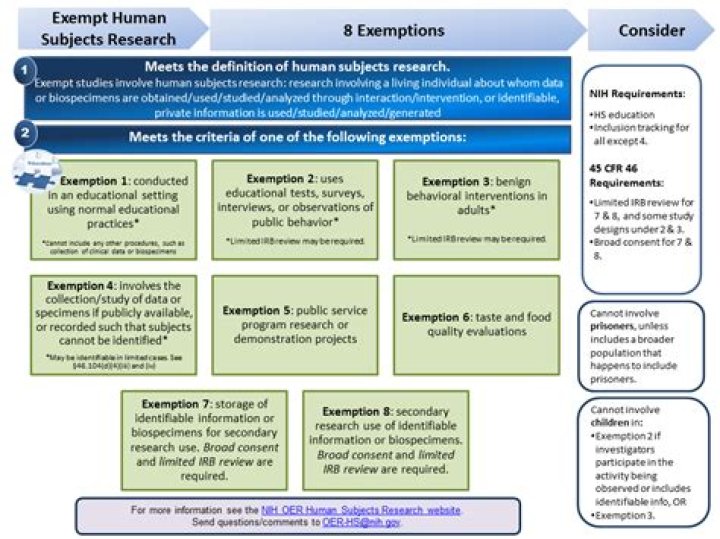
Exempt studies involve human subjects research: research involving a living individual about whom data. or biospecimens are obtained/used/studied/analyzed through interaction/intervention, or identifiable, private information is used/studied/analyzed/generated.
What does exempt IRB status mean?
The categorization of human subjects research as “exempt” from IRB review or appropriate for “expedited” IRB review is intended to streamline IRB procedures with no diminution of protection for human subjects.
What are the three types of IRB review?
There are three major types of review: Exempt, Expedited, and Full.
What does IRB exempt status mean?
When would you use an IRB?
FDA regulations generally require IRB review and approval of research involving FDA-regulated products (e.g., investigational drugs, biological products, medical devices and dietary supplements) (21 CFR Part 56).
What is exempt from IRB?
Who determines if research is exempt from federal regulations?
Who may determine that research is exempt? The regulations do not specify who at an institution may determine that research is exempt under 45 CFR 46.101(b).
How do I know if I am exempt or nonexempt?
Most employees must meet all three “tests” to be exempt. Salary level test. Employees who are paid less than $23,600 per year ($455 per week) are nonexempt. (Employees who earn more than $100,000 per year are almost certainly exempt.)
What does the IRB look for?
The IRB needs to be assured that the research is 1) of sound design, given the proposed use of human subjects; 2) that there is equitable selection of subjects; 3) that there is a reasonable balance in the risks and benefits to the participants; and, 4) that the informed consent process is appropriate and comprehensive …
Does qualitative research need IRB approval?
All non-exempt research involving human subjects must have advance IRB approval. Qualitative research often does not need IRB, Meyer said.